Phenylacetylene (C8H6)
This shows different orbitals and charge differences for the
molecule nitrogen dioxide. There are also different vibrations for this
molecule toward the bottom. The best Dipole Moment we have for this
molecule is at 0.697708 Debye while the theoretical was 0.656 Debye.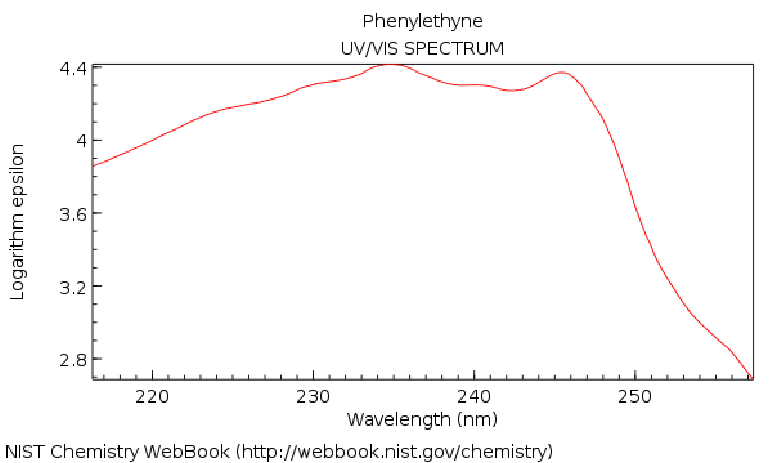
| Transition from Ground State to Excited State # |
Oscillator Strength |
Excitation Energy (nm) |
| 1 |
0.204532 |
210.17 |
| 2 |
0.001935 |
203.17 |
| 5 |
0.093386 |
174.26 |
| 6 |
0.926358 |
154.25 |
| 7 |
1.783207 |
150.39 |
| 9 |
0.004835 |
131.58 |
| 10 |
0.006934 |
126.47 |
IR Spectrum
C8H6 Geometries
Vibration 1-10
Vibration 11-20
Vibration 21-36
Comparing the calculated vibrations to the IR Spectrum, we get the following;
vibrations 1-2 are too small to be in the IR Spectrum shown
first semi-large peak on right ~550cm-1 contains vibrations 3-6
second large peak on right ~600-750cm-1 contains vibrations 7-9
third semi-large peak on right ~800cm-1 contains vibrations 10-13
fourth semi-large peak on right ~950cm-1 contains vibration 14
fifth small dual peaks on right ~1000-1150cm-1 contains vibrations 15-20
sixth semi-large peaks on right ~1250-1400cm-1 contains vibrations 21-25
seventh semi-large triple peaks on right ~1450-1700cm-1 contains vibrations 26-29
eighth semi-small peak on right ~2200cm-1 contains vibration 30
second large peak on left ~3100 contains vibrations 31-35
first large peak on left ~3300 contains vibration 36
Highest Occupied Molecular Orbital (HOMO). HOMO
was calculated using valence electrons from both atoms and divided by 2
in order to represent that there are two electrons on each sub-shell.
| ||||||
Lowest Unoccupied Molecular Orbital (LUMO). After the HOMO state, LUMO
represents when one electron has entered into the next excited state.
| ||||||
Electrostatic potential is the potential for electrons to be in certain
places around the molecule. The red represents a very likely place for
the electrons, while going down the color spectrum to blue is where the
electrons are not likely to be around.
| ||||||
Partial atomic charges relates to the Molecular Electrostatic Potential
in that there is an uneven charge distribution. Partial atomic charges
gives the specific non-integer values that shows the polarity.
|
Page skeleton and JavaScript generated by export to web function using Jmol 14.2.12_2015.01.22 2015-01-22 21:48 on Mar 3, 2015.