Theoretical Calculations of Properties of Toluene
by Dustyn Sawall
Types of calculations performed
In general, Molecular orbitals (MO) are combinations of Atomic
orbitals (AO). Atomic orbitals are actually mathematical expressions
representing where a electron in a given quantum state can be found.
When AO's are added together, they are done so in a way to minimize
the energy of the system. This is done by some contributions of AO's
being greater than others. The mathematical formula for this
simplifies into three type of factors, the energy of the original AO,
the energy saved by forming the bond and an overlap factor dependent
on multiple interactions between electrons that need to be evaluated
at the same time.
These equations rapidly become huge. To do them for all of the
overlap considerations takes considerable time and computing power.
Even then, they don't all work out. These are known as ab initio
calculations. To improve workability of the mathematics, some
modifications can be made. Generally, the more of the overlap
integrals you use, the better your calc, but that is a trade off with
if they can be done at all.
In the programs used for this page, three different levels of
theory are analyzed. In the extended Huckel theory, it is assumed
that only nearest neighbors interact. Zindo level of theory uses
intermediate neglect of the overlap intergrals. It works using some
empirical data from ab initio calculations to optimize its MO's.
MOPAC level of theory uses a moderate neglect of these intergrals and
uses empirical data from thermodynamics to improve it's theory. MOPAC
is the most precise of the programs used because it takes into
account more of the intergrals in question.
Results of geometry optimization at various levels of theory
Initially, only a Molecular Mechanics program was used. This
program treats the atoms as classical harmonic oscillator. The bond
lengths and angles are calculated from rotational and vibrational
spectroscopy results. The computer has tables of bond lengths, the
computer then uses standard values from these tables to optimize the
molecules. The molecule is designed so as to minimize the stress
between atoms. This is the program used for the extended huckel level
of theory. Zindo and MOPAC have geometry optimizations available to
them from quantum theory to further optimize the molecules. In
theory, the Mopac calc should be best. If the bond lengths and
angles were examined in detail, there would be slight differences,
but as can be seen from the pictures below, they are slight.
The molecular orbitals that provide the primary bonding
Here are a few of the molecular orbitals for toluene. This is only
two of the orbitals that provide bonding for the molecule, there are
several others. As can be seen, the MO's can cover large portions of
the molecule.
|
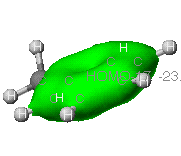
This is an example of a sigma bond from the carbon atoms
throughout the benzene ring.
|
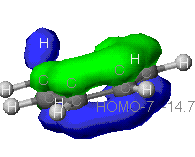
This is one veiw of the pi bond formed from the p orbitals
in the carbons of the benzene ring.
|
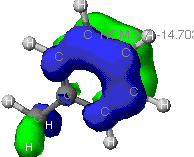
This is simply another veiw of the pi bond. Notice how the
orbital extends in to the co-planar H's of the methyl
group.
|
The highest occupied (HOMO) and lowest unoccupied (LUMO)
molecular orbitals
These are the orbitals that are most involved in chemical
reactions since they can most easily lose or gain electrons.
|
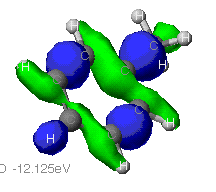 Extended Huckel Extended Huckel
|
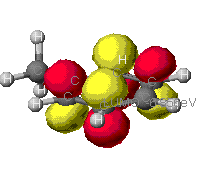
|
|
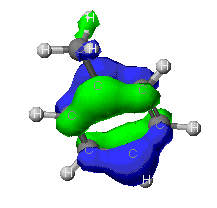 MOPAC
and ZINDO MOPAC
and ZINDO
|
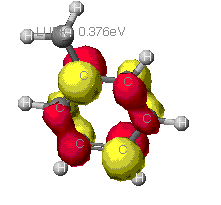
|
As can be seen from these diagrams, the extended Huckel
theory and the other two often differ. This is because of the
different assumptions each make. In general the MOPAC level of theory
is superior. These show the orbitals that are at the Highest Occupied
MO, that is the MO that is occupied at an elevated energy level. The
red/yellow is the Lowest Unoccupied MO. There are no electrons in
these MO's as they are at such an elevated energy level.
Electron density/distribution maps
|
 MOPAC MOPAC
|
This is diagram showing the probability of finding an
electron in the given areas of map, the warmer the color, the more
likely that an electron could be found there.
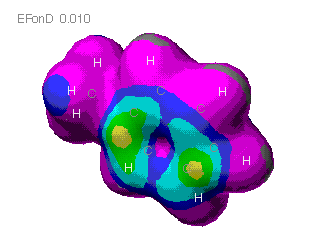
Charge distribution maps
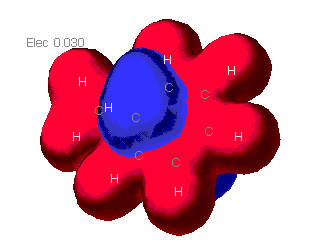 MOPAC
MOPAC
These maps show the relative distribution of positive and negative
charge on the molecules. The red indicates a positive charge and blue
is for a negative charge. As can be seen here, the molecule mostly
presents a positive charge uniformally. The only negative areas are
outside of the plane of the molecule. It would be predicted that a
partial negative charge would be found above and below the ring. A
partial positve charge could be predicted near the methyl group.
Calculated dipole moments
|
Method used
|
Dipole Moment
|
|
Extended Huckel
|
.733
|
|
Mopac
|
.262
|
|
Zindo
|
.525
|
|
CRC
|
.375
|
Sep_Lab_Home.html to
return to the main page



 Extended Huckel
Extended Huckel
 MOPAC
and ZINDO
MOPAC
and ZINDO

 MOPAC
MOPAC
 MOPAC
MOPAC