Dichloromethane
The calculated bond lengths
and angles for the 6-21G calculations are shown below. This was the
lowest level of theory used to calculate molecular geometry.6-31G was the next highest level of theory used to calculate bond length and bond angles. This level of theory generated bond lengths and angles that most closely matched the literature8 values presented in Table 1 and Table 2.
DZV was the highest level of theory used to calculate molecular geometry. Buttons showing the calculate bond length and bond angle are presented below.
Table 1: Literature values for the angles between atoms on dichloromethane.
| Reference Atoms |
Angle Between Atoms (o) |
| C-H |
112 |
| C-Cl |
108.275 |
| H-H |
112 |
| Cl-Cl |
111.785 |
Table 2: Literature values for the lengths of bonds comprising dichloromethane.
| Type of Bond |
Length of Bond (nm) |
| C-H |
0.10680 |
| C-Cl |
0.17724 |
The calculated partial atomic charges for dichloromethane are shown below. These values represent the distribution of electrons between atoms.
The highest occupied molecular orbital (HOMO) is shown below. This is the orbital geometry that represents the valence electrons in their ground state. This was calculated by summing the total number of electrons for the molecule and dividing by two.
The lowest unoccupied molecular orbital (LUMO) is show below. This is the orbital that the valence electrons occupy once they have been excited by some form of energy. This was calculated by selecting the molecular orbital that was adjacent to the HOMO orbital but with a differing sign associated with the energy.
The electrostatic potential graphic shown below, represents the probability density of where the bonding electrons are located in a given molecule. Red colors represent regions of greater probability density, while blue colors represent regions of lower density. Colors between red and blue represent intermediate probability densities.
Graphics representing the vibrational frequencies of dichloromethane are shown below. All of these vibrational frequencies were calculated using the DZV level of theory as it offered vibrational frequencies closest to that of the literature8 values. Vibrational frequencies for molecules are most often observed through IR spectra. An IR spectra for dichloromethane is shown below as Figure 1.
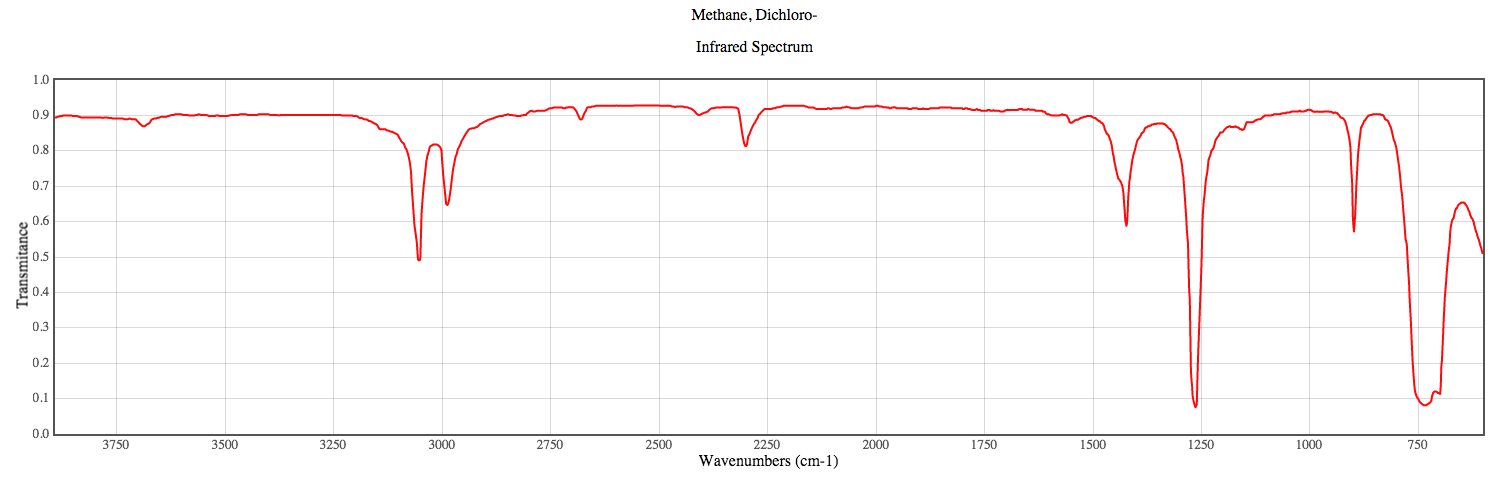
Figure 1: A representative IR spectra of dichloromethane.
All of the vibrational models shown are representative of major peaks shown in Figure 1.
The vibration at 1283.99 cm-1 represents a C-C bend.
The vibration at 1424.41 cm-1 represents a C-H bend.
The vibration at 3393.11 cm-1 represents a C-H stretch.
The dipole moment for dichloromethane was calculated at each level of theory and compared to the literature8 value of 1.600 debyes. The DZV model of dichloromethane gave the closest value of 1.8680, giving an error of 16.75%. The DZV model should have produced the closest dipole moment, as this level of theory uses the greatest number of basis sets.
You may look at any of these intermediate views again by clicking on the appropriate button.
Page skeleton and JavaScript generated by export to web function using Jmol 14.2.12_2015.01.22 2015-01-22 21:48 on Mar 2, 2015.
This will be the viewer

If your browser/OS combination is Java capable, you will get snappier performance if you use Java