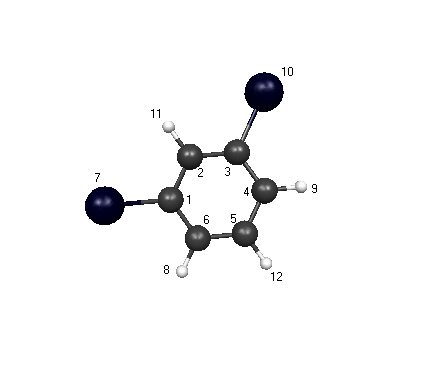
geometry due to the fact that it gave the highest
correlation between the literature values of the
bond lenghts and dipole moments and the experimental values calculated.
| Atom 1 |
Atom 2 |
Length |
| 1 |
2 |
1.380463 |
| 1 |
6 |
1.380383 |
| 1 |
7 |
1.927641 |
| 2 |
3 |
1.380486 |
| 2 |
11 |
1.069065 |
| 3 |
4 |
1.350377 |
| 3 |
10 |
1.927639 |
| 4 |
5 |
1.387061 |
| 4 |
9 |
1.070407 |
| 5 |
6 |
1.387061 |
| 5 |
12 |
1.072079 |
| 6 |
8 |
1.070389 |
Dipole Moment: 1.784369 Debye
Literature Value for dibromobenzene: 1.44 Debye
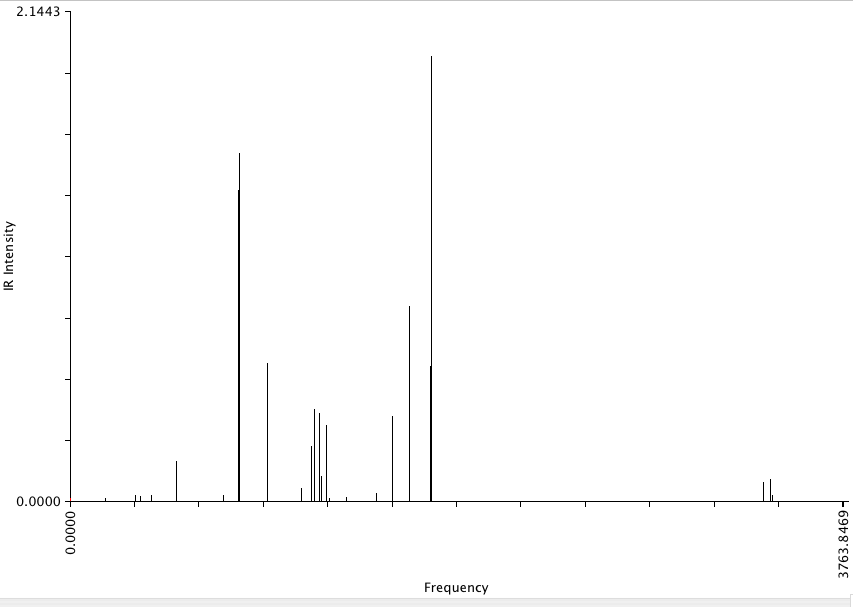
frequencies for meta-dibromobenzene.
The large peaks at 1758.79, 1650.80, 827.71, 818.08 cm-1 represent the vibrational modes of the C-C bonds in the aromatic ring.
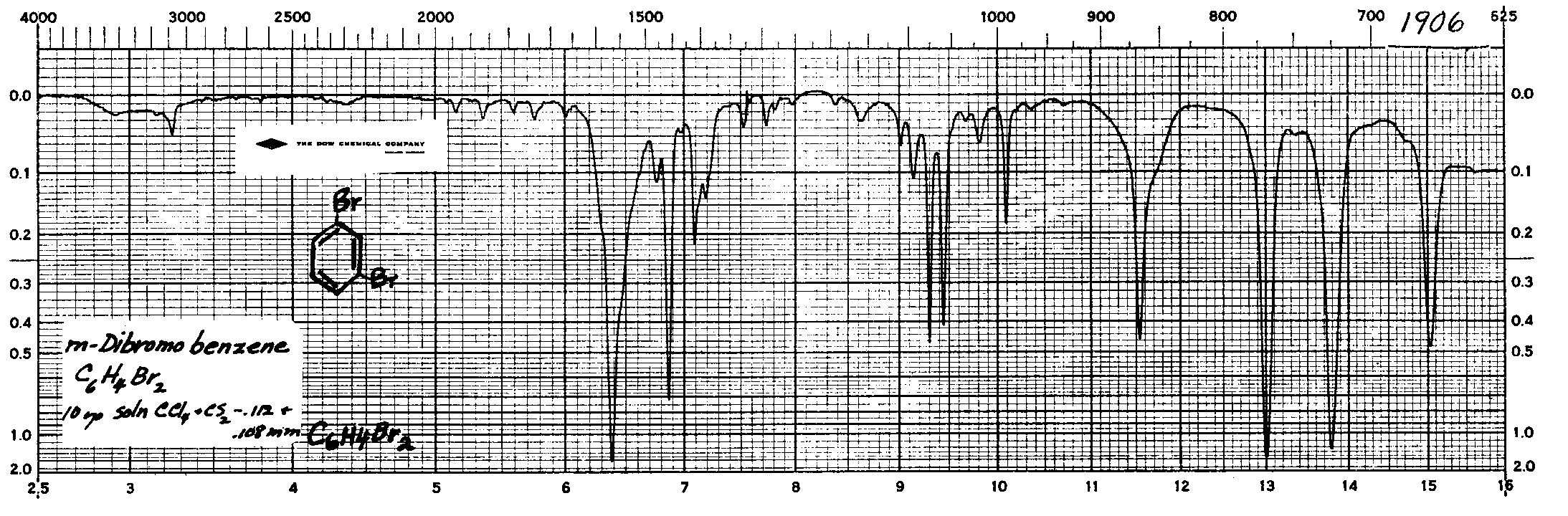
Peaks representing the C-C vibrational modes appear in the spectrum at 1625, 1400, 770, 730 cm-1