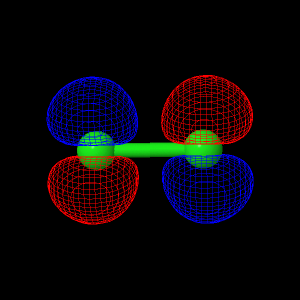
Dichlorine Quantum Calculations
All of the live displays on this page were created using the best optimized geometry. The best found geometry optimization was at the ab initio theory level of 6-31G.
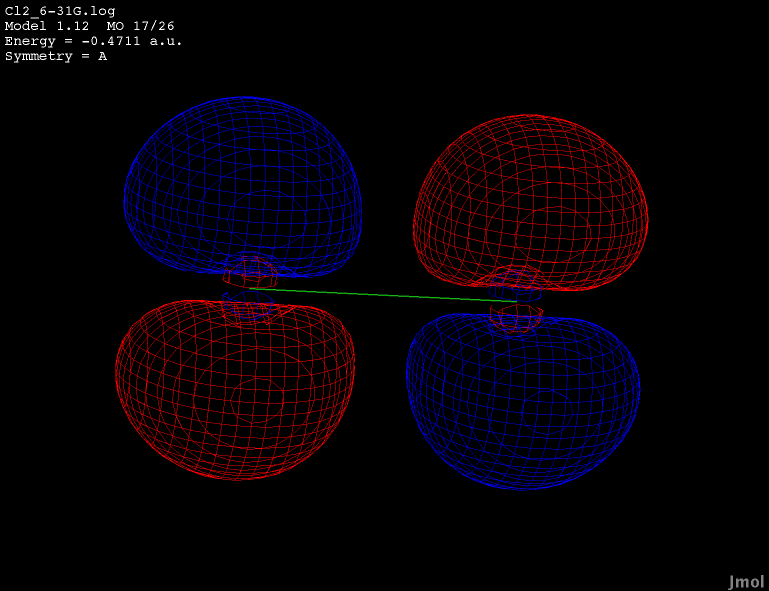
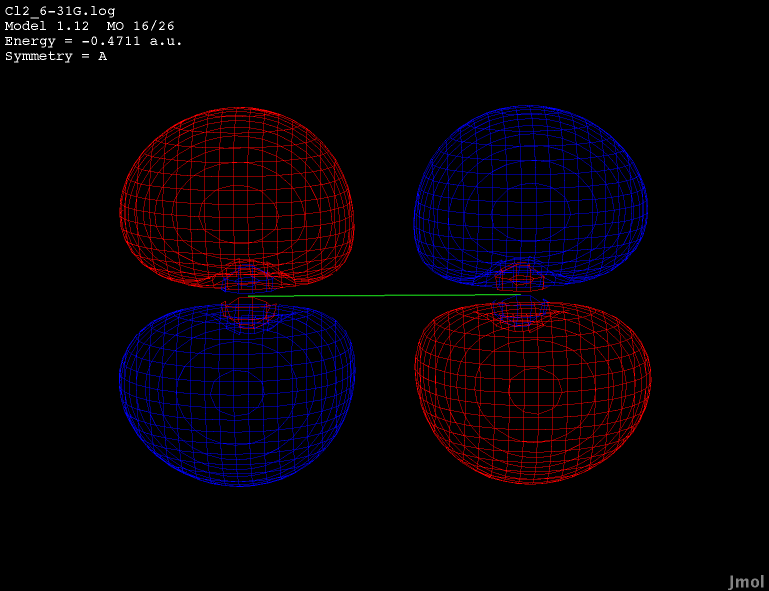
Dichlorine has doubly degenerate HOMO orbitals. Both MO16
and MO17 show the same energy of -0.4711a.u. All of the live displays on this page were created using the best optimized geometry. The best found geometry optimization was at the ab initio theory level of 6-31G.
The button below shows one of the HOMO orbitals for dichlorine.
|
|
The button below shows the second HOMO orbital for dichlorine.
|
|
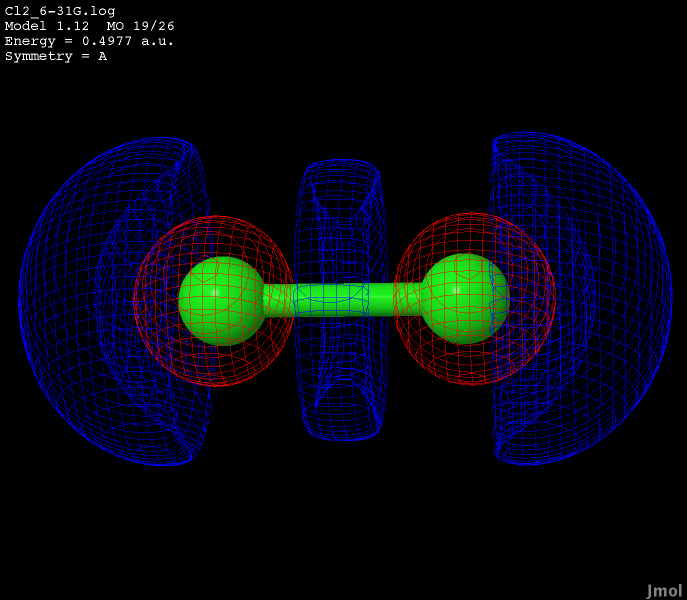
The LUMO orbital of dichlorine is found at an energy of -0.0357a.u.
Clicking the button below will show a live display of the LUMO orbital.
|
|
Below is a table of molecular orbitals showing the shell number, occupancy, and bonding status. The table is set up similarly to an energy level diagram.
 |
4s orbital - unoccupied bonding |
 |
3p orbital - unoccupied LUMO anti-bonding |
 |
3p orbital - occupied HOMO anti-bonding Major Contributor |
 |
3p orbital - occupied bonding Major Contributor |
 |
3p orbital - occupied anti-bonding Major Contributor |
 |
3p orbital - occupied bonding Major Contributor |
 |
3p orbital - occupied bonding Major Contributor |
 |
3s orbital - occupied anti-bonding |
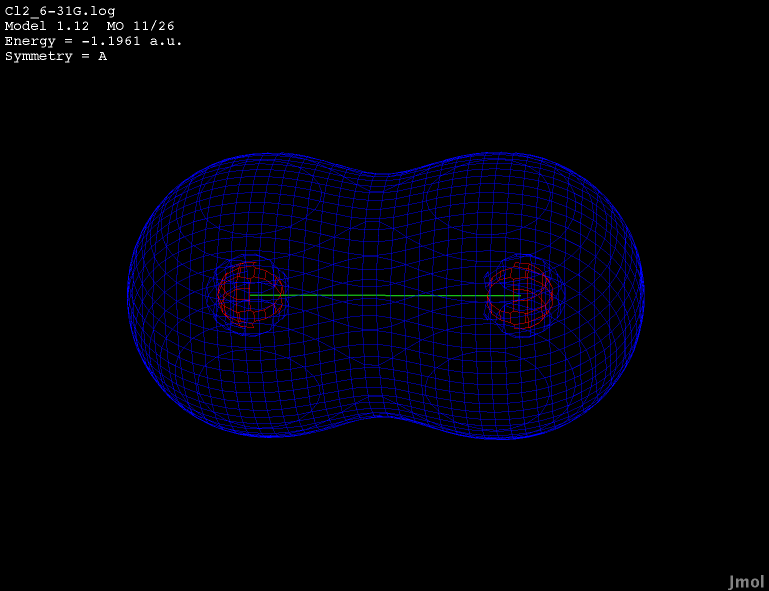 |
3s orbital - occupied bonding |
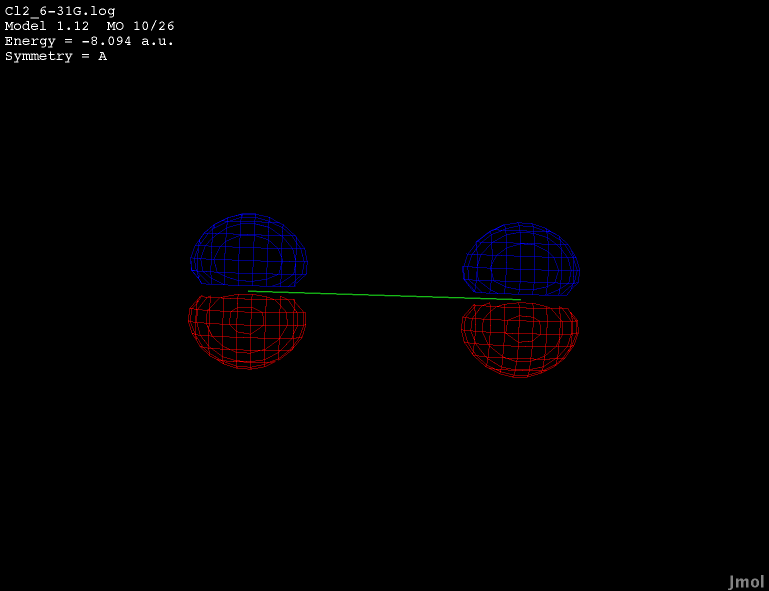 |
2p orbital - occupied bonding |
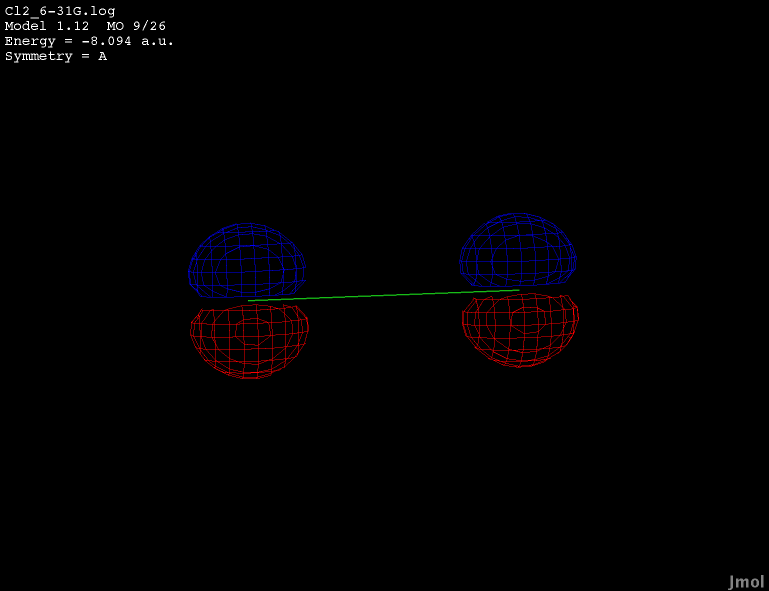 |
2p orbital - occupied bonding |
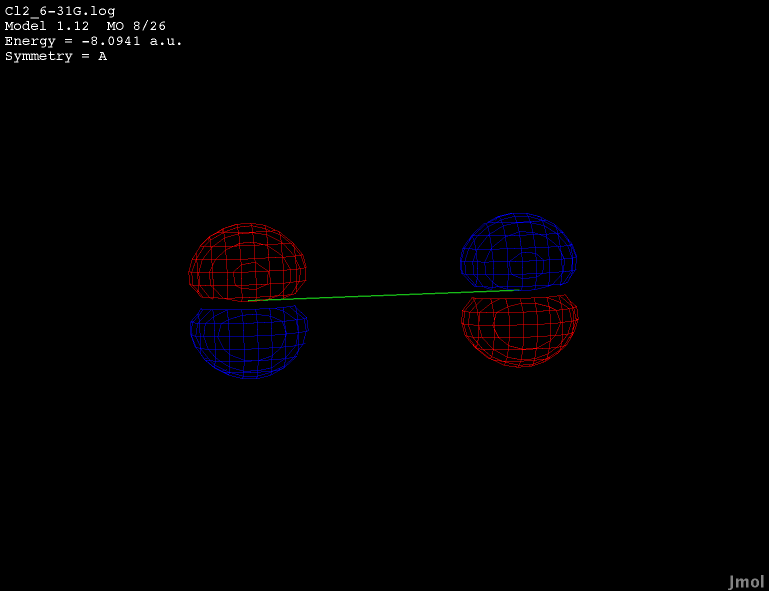 |
2p orbital - occupied anti-bonding |
 |
2p orbital - occupied anti-bonding |
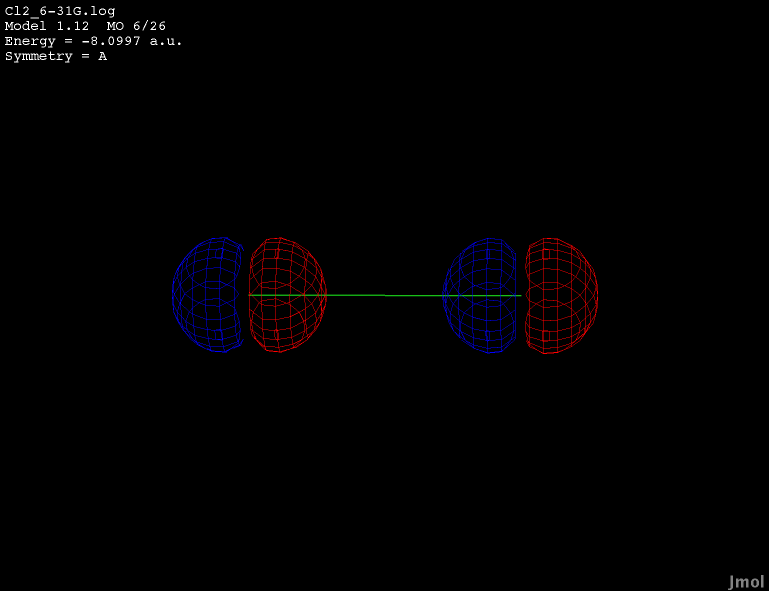 |
2p orbital - occupied anti-bonding |
 |
2p orbital - occupied bonding |
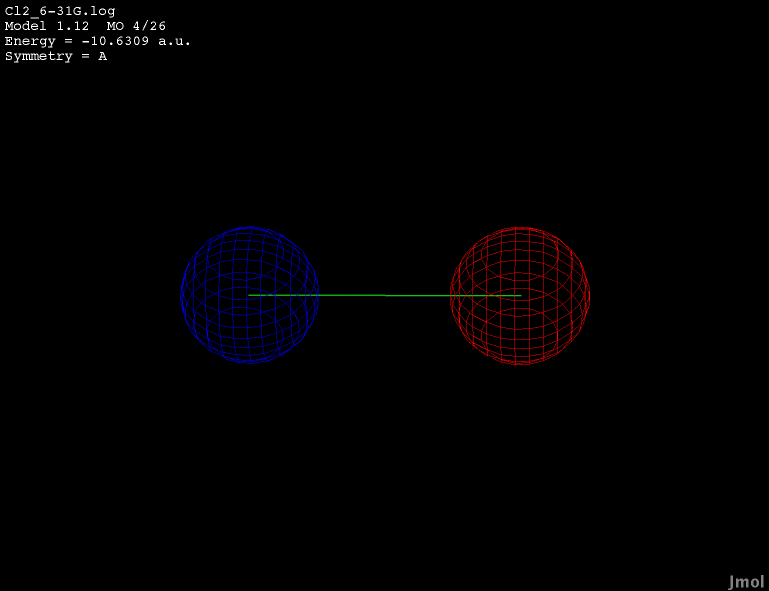 |
2s orbital - occupied anti-bonding |
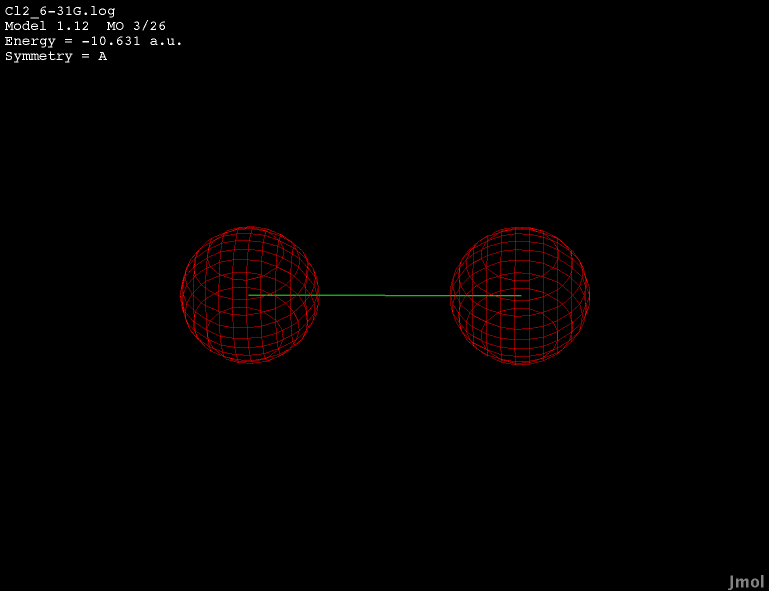 |
2s orbital - occupied bonding |
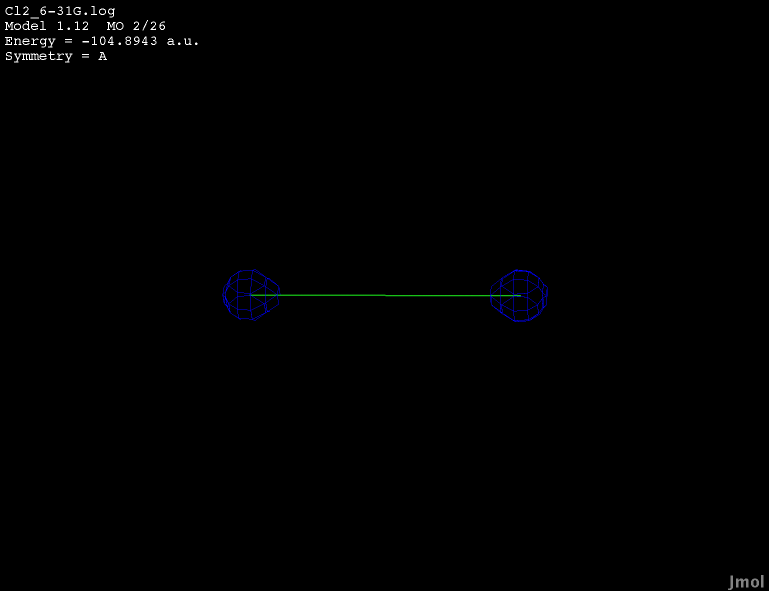 |
1s orbital - occupied bonding |
 |
1s orbital - occupied anti-bonding |
|
|
The potential energy of bond stretching for the 6-31G basis set was found to be the lowest, which also aided in the selection of the best basis set.
The button below shows a live display of the dichlorine molecule in its most optimal basis set of 6-31G.
|
|
A plot of the potential energy of bond stretching at different levels of theory can be seen below.7
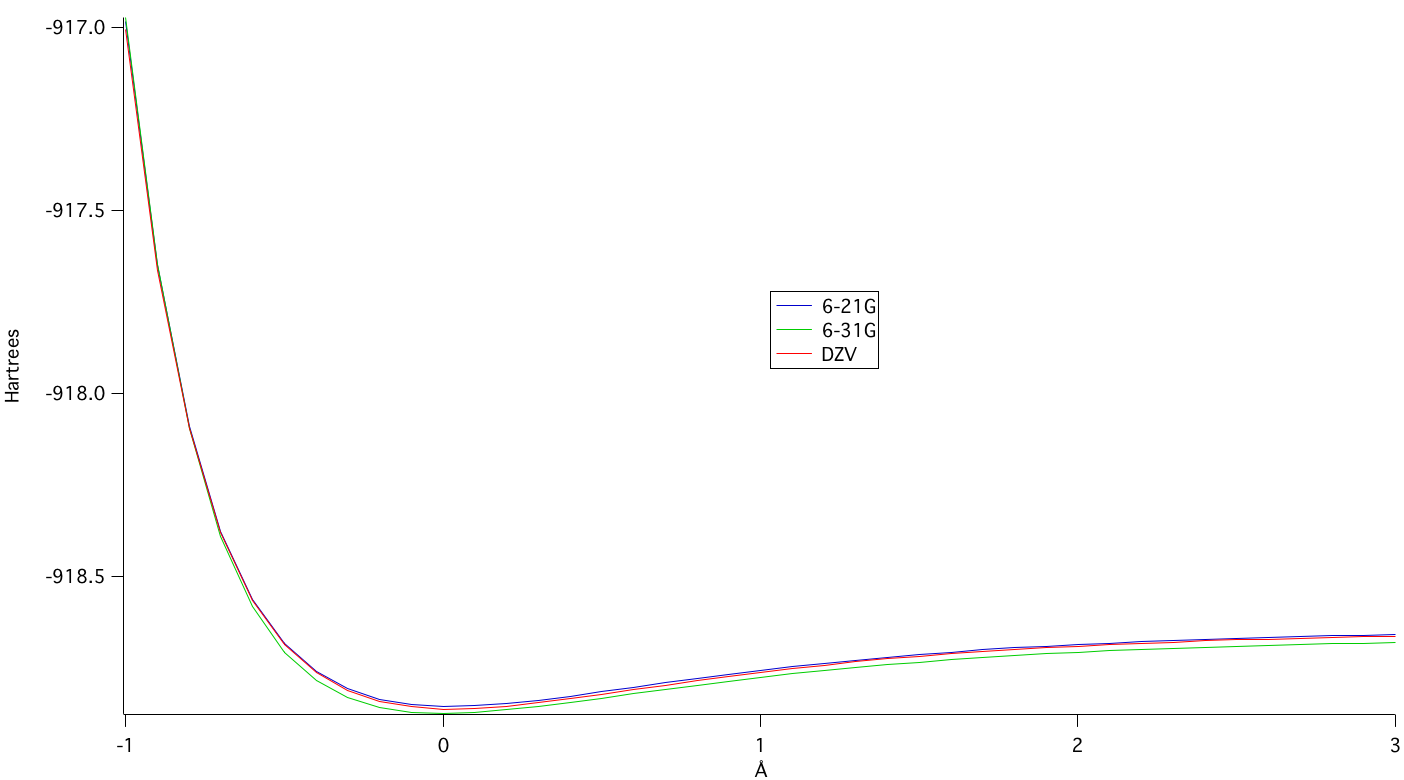
The potential energies of the basis sets go from: 6-31G > DZV > 6-21G
The vibrational frequency of the molecule was found using the DZV basis set. The value of the vibrational frequency was found to be 516.80cm-1.
Dichlorine has no dipole because it is a homonuclear diatomic molecule.
Dichlorine will not have partial charges because it is a homonuclear molecule.
There will be no electrostatic potential because the electrons in this diatomic molecule are evenly distributed.
References
Page
skeleton and JavaScript generated by export to web function
using Jmol 12.2.34
2012-08-09 20:37 on Feb 26, 2013.