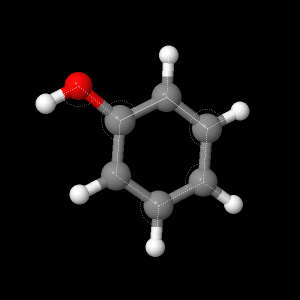
Below shows the structure of the calculated phenol. Take note of how the structure is set up to promote lowest possible energy; such as the uniform ring and the oxygen having different bond angles from the Hydrogens on the ring to compensate for the non-linear OH.
|
|
The highest occupied molecular orbital showing the 2p orbitals
|
|
The lowest unoccupied molecular orbital showing the 2p anti-bonding orbitals
|
|
Electrostatic Potential and Labeled Partial Atomic Charges.
|
|
Further Information:
Excited State Energies of Phenol
Vibrational Energies of Phenol
Page
skeleton and JavaScript generated by export to web function
using Jmol 12.2.34
2012-08-09 20:37 on Feb 26, 2013.