Chlorobromide
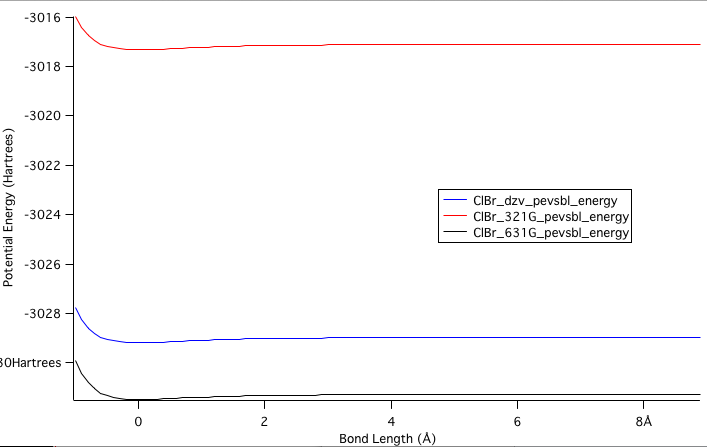
Below shows a graph of Potential Energy vs Bond Length, which compares the different calculated molecules that were made in different software. The 3-21G line was the lowest calculated molecule, so it is no surprise that it is the highest potential energy, which means that this is the worst data of the three. The 6-31G was calculated next, which provided our best calculated value according to this graph, which is surprising since the DZV calculation had the most calculations done to it. For this reason, all later calculations were done using the 6-31G calculation.
Chlorobromide Geometries
The best calculated Dipole Moment 1.074637 Debye was while the theoretical Dipole Moment was 0.519 Debye.
MO Diagram
Highest Occupied Molecular Orbital (HOMO). There are two different HOMO
states for the molecule due to the degeneracy of the excited state. HOMO
was calculated using valence electrons from both atoms and divided by 2
in order to represent that there are two electrons on each sub-shell.
| ||||||
Lowest Unoccupied Molecular Orbital (LUMO). After the HOMO state, LUMO
represents when one electron has entered into the next excited state.
| ||||||
Electrostatic potential is the potential for electrons to be in certain
places around the molecule. The red represents a very likely place for
the electrons, while going down the color spectrum to blue is where the
electrons are not likely to be around.
| ||||||
Partial atomic charges relates to the Molecular Electrostatic Potential
in that there is an uneven charge distribution. Partial atomic charges
gives the specific non-integer values that shows the polarity. Since
this molecule has only two atoms, the numbers will be equal values but
opposite signs (+-).
| ||||||
This vibration is the only vibration this molecule has because there are
only two atoms. Since the diatomic has only one mode of vibration based
on 3N-5. The vibrational frequency was at 417.01cm-1.
|
Page skeleton and JavaScript generated by export to web function using Jmol 14.2.12_2015.01.22 2015-01-22 21:48 on Mar 2, 2015.