Once the molecule file is fully loaded, the image at right will become live. At that time the "activate 3-D" icon ![]() will disappear.
will disappear.
Dye Molecule
1,1-diethyl-2,2 cyanine
This complex looking molecule is a well known dye most commonly
used in silver halide photography and has a red or pinkish look to it
when in solution. This molecule was of interest because of its
conjugated chain connecting the two halves of the molecule. This not
only allows for it to have interesting light absorbing characteristics,
but as a consequence of its affinity for absorbing a specific wavelength
of light, offers multiple ways to measure the bond distance between
carbons in the conjugated chain.
Click the button below to see the electrostatic potential mapping of this molecule.
The electrostatic mapping of this molecule was done using the CCD calculations obtained from the molecular geometry calculations. You can see areas with higher electron density highlighted in warmer colors like red, and lower electron density in cooler colors like blue.
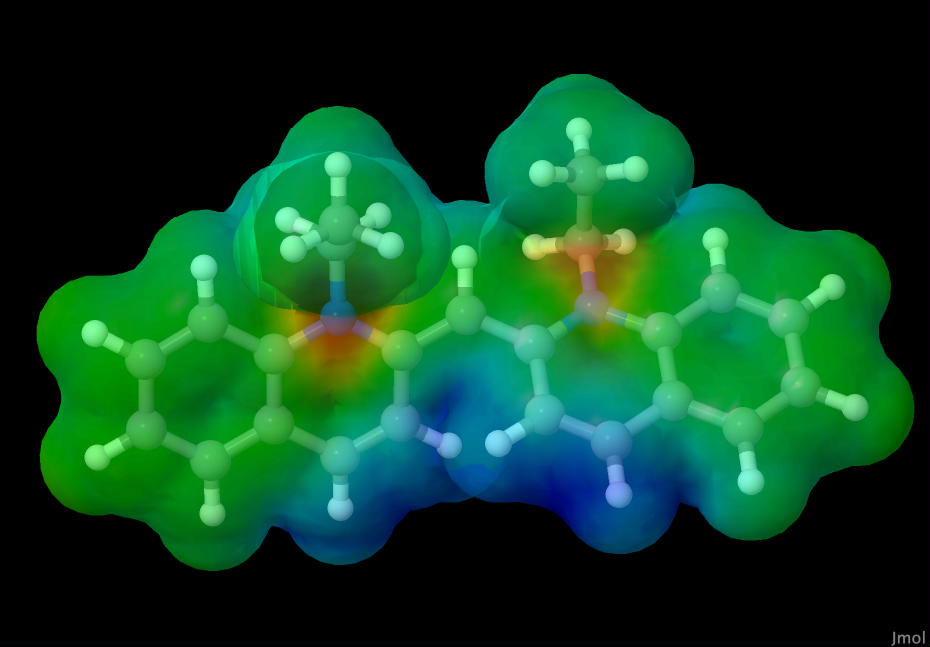 |
| Dye molecule's electrostatic potential map |
The electrostatic mapping of this molecule was done using the CCD calculations obtained from the molecular geometry calculations. You can see areas with higher electron density highlighted in warmer colors like red, and lower electron density in cooler colors like blue.
Click the button below to see the highest occupied molecular orbital.
The highest occupied molecular orbital or HOMO for short, shows the outer most orbitals that electrons will occupy when the molecule is in the ground state. Being the outer most shells, this also means that this is were the highest energy electrons will be found. These calculations were made determining the total number of electrons in the molecule, and dividing it by 2, since each molecular orbital can hold 2 electrons with opposite spin. In this case the HOMO ended up being the 87th out of 97 total calculated molecular orbitals.
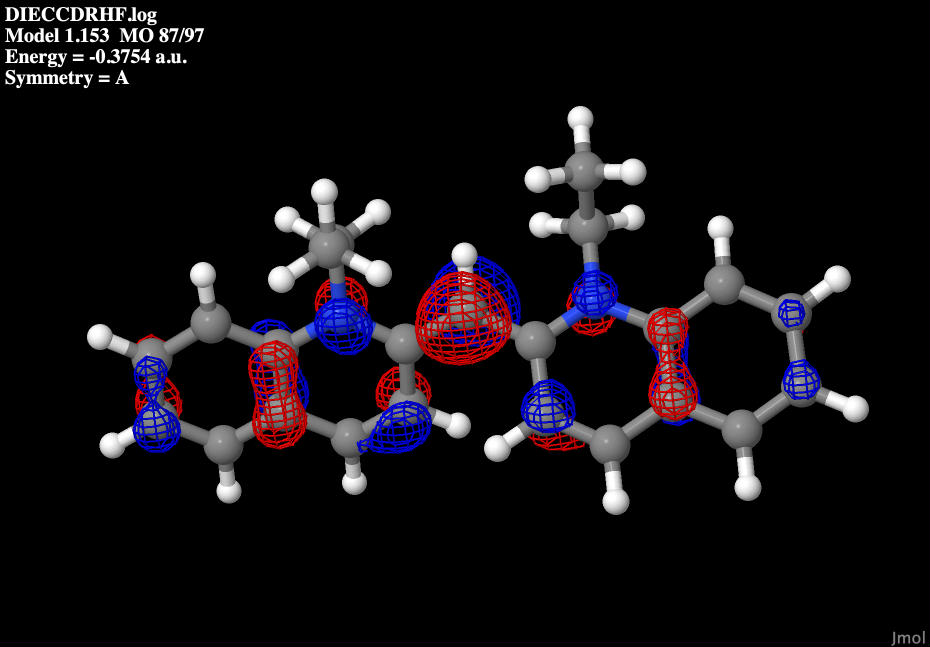 |
| Dye molecule's HOMO |
The highest occupied molecular orbital or HOMO for short, shows the outer most orbitals that electrons will occupy when the molecule is in the ground state. Being the outer most shells, this also means that this is were the highest energy electrons will be found. These calculations were made determining the total number of electrons in the molecule, and dividing it by 2, since each molecular orbital can hold 2 electrons with opposite spin. In this case the HOMO ended up being the 87th out of 97 total calculated molecular orbitals.
Click the botton below to see the lowest unoccupied molecular orbital
The lowest unoccupied molecular orbits, or LUMO for short, are the orbitals that are just higher in energy than the HOMO, in this case the 88th molecular orbital. These are the first orbitals that electrons will move to when the molecule is excited.
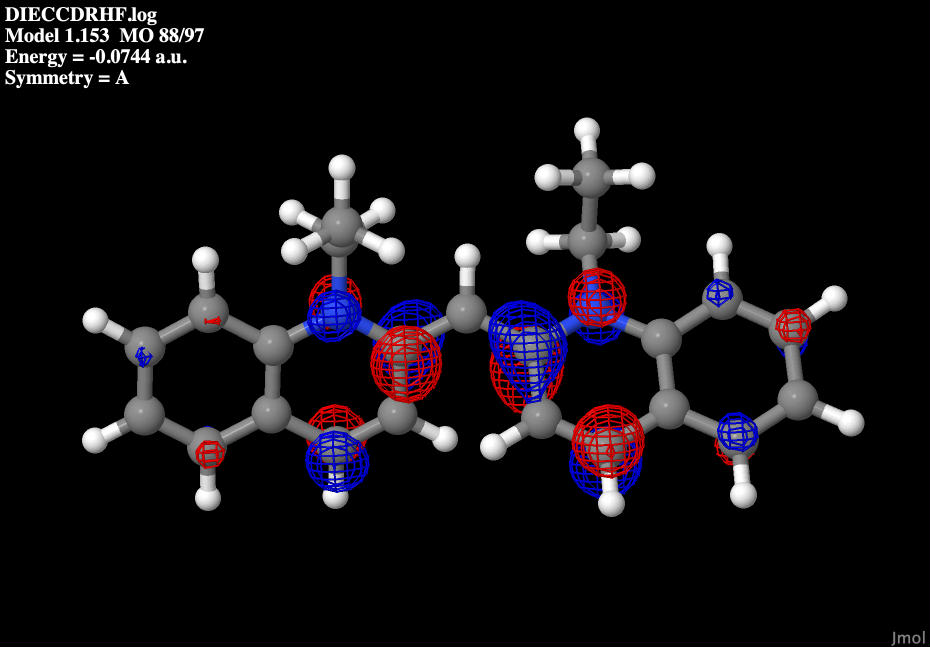 |
| Dye molecule's LUMO |
The lowest unoccupied molecular orbits, or LUMO for short, are the orbitals that are just higher in energy than the HOMO, in this case the 88th molecular orbital. These are the first orbitals that electrons will move to when the molecule is excited.
Click the button below to see the bond angle
As you can see, the CCD calculations began to find the geometrically lowest energy state for the molecule to be in. This has manifested itself as a twisting around the central carbon atom. Although lower energy states are likely for this molecule, after 150 instances, the geometry that the GamessQ software has calculated is a 75.8 degree twist between the two sides of the molecule
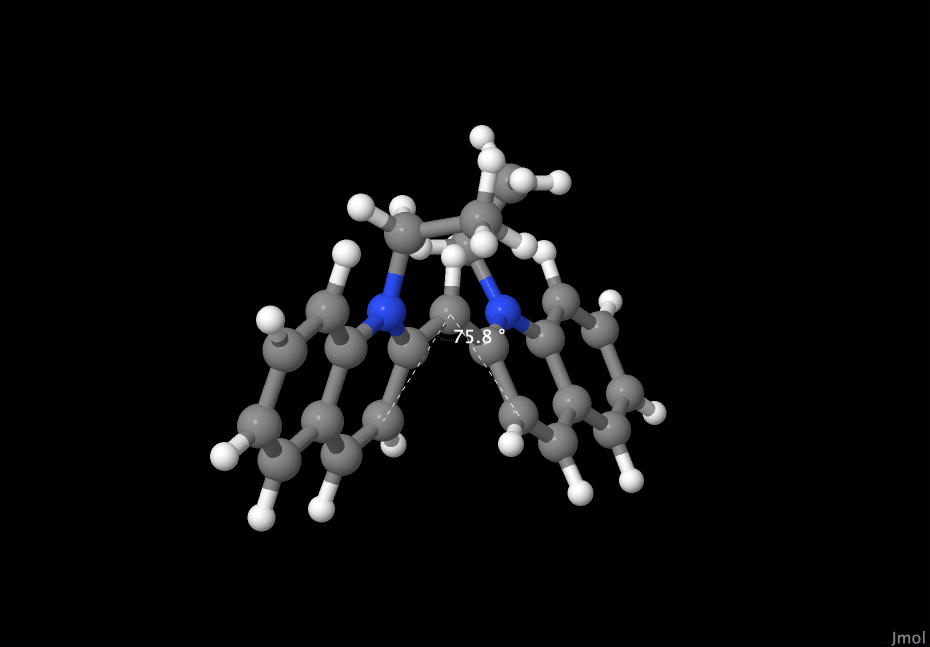 |
| Dye molecule's bond angle |
As you can see, the CCD calculations began to find the geometrically lowest energy state for the molecule to be in. This has manifested itself as a twisting around the central carbon atom. Although lower energy states are likely for this molecule, after 150 instances, the geometry that the GamessQ software has calculated is a 75.8 degree twist between the two sides of the molecule
