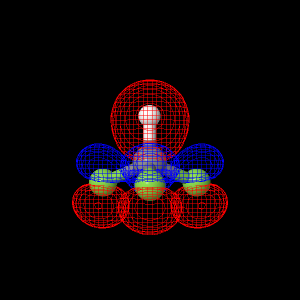
Trifluoromethane Molecular
Orbital Calculations
The Highest Occupied Molecular Orbital (HOMO) for
Trifluoromethane (CHF3) was determined using the
Double Zeta Valence (DZV) level of theory. |
|
The Lowest Unoccupied Molecular Orbital (LUMO) for CHF3 was based off of DZV calculations.
|
|
Th electrostatic potential of CHF3 was mapped; red represents high electrons density and blue represents low electron density.
|
|
The partial atomic charges for carbon, hydrogen, and fluorine in CHF3 were determined based on the DZV level of theory.
|
|
The bond lengths and bond angles for CHF3 were determined based on the DZV geometry optimization.
|
|
Table 1: Experimental and literature values for bond lengths and angles for CHF3.1
| Bond |
Bond Length (Å) |
Bond Angle (º) |
| C-H |
1.098±0.008 |
--------------------- |
| C-F |
1.332±0.008 |
108.8 |
An IR spectrum for CHF3 can be seen by following the link below.
CHF3 IR Spectrum
Table 2: The wavenumbers and the corresponding bending and/or stretching that occurs at the significant peaks on the IR spectra above.2
| Wavenumber (1/cm) |
Bond and Motion |
| ~1150 |
C-F stretching |
| ~1400 |
Csp3-bending |
| ~3050 |
Csp3-H stretching |
The button Fluorine Bending Mode will appear in the box below. Insert information for Fluorine Bending Mode here and below.
|
|
The button Fluorine Bending and Stretching will appear in the box below. Insert information for Fluorine Bending and Stretching here and below.
|
|
The button Hydrogen Bending will appear in the box below. Insert information for Hydrogen Bending here and below.
|
|
The button Hydrogen Stretching will appear in the box below. Insert information for Hydrogen Stretching here and below.
|
|
Table 3: The dipole moments of CHF3 at the different levels of theory.1
| Level of Theory |
Dipole Moment (D) |
| AM1 |
2.079345 |
| 6-31G |
2.372354 |
| 6-21G |
2.106802 |
| DZV |
2.554768 |
| Theoretical |
1.651 |
Page
skeleton and JavaScript generated by export to web function
using Jmol 12.2.34
2012-08-09 20:37 on Feb 25, 2013.