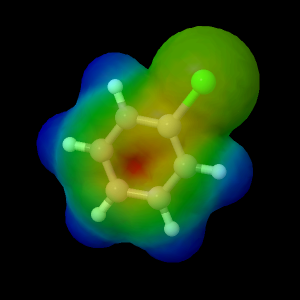
Chlorobenzene Molecular
Orbital Calculations
The electrostatic potential of chlorobenzene (C
6H
5Cl)
was determined using the Double Zeta Valence (DZV) level of
theory.
The partial atomic charges for carbon, hydrogen, and chlorine in
C
6H
5Cl were determined based on the DZV
calculation.
The bond lengths and bond angles of C
6H
5Cl
were determined based on the DZV geometry optimization.
The DZV level of theory was chosen for this molecule because it
determined the most accurate bond lengths and bond angles in
comparison with the literature.
Table 1: Literature values for bond lengths and angles
for C
6H
5Cl.
1
Bond
|
Bond Length (Å) |
Bond Angle (º) |
C-H
|
1.084±0.006 |
120º |
C=C
|
1.395±0.003 |
120º |
C-Cl
|
1.810±0.004 |
120º |
An IR spectrum for C
6H
5Cl can be seen by
following the link below.
C6H5Cl
IR Spectrum
Table 2: The wavenumbers of the significant peaks located
on the IR Spectra above.
2
Wavenumber (cm-1)
|
Bond
|
~750
|
C-Cl stretch
|
~1100
|
Csp2-H bend
|
~1500
|
C=C bend/stretch
|
~1600
|
C=C stretch
|
~3100
|
Csp2-H stretch
|
Below are the different frequencies at which the different bonds
stretch and bend in an IR Spectrum.
The Highest Occupied Molecular Orbital (HOMO) for C
6H
5Cl
was calculated using the DZV level of theory.
The Lowest Unoccupied Molecular Orbital (LUMO) for C
6H
5Cl
was determined using DZV.
Table 3: The literature and experimental values of the
dipole moments for C
6H
5Cl.
1
Calculated
|
1.306130D
|
Theoretical
|
1.690D |
The value calculated using AM1 was the most accurate calculated
dipole moment.
Table 4: DZV level of theory UV-Vis
Absorption Peaks.
Ground State to Excited State #:
|
Energy (cm-1)
|
2
|
52512.90
|
3
|
61100.79
|
4
|
67323.93 |
5
|
67845.29 |
6
|
73131.68
|
9
|
78495.30
|
The numbers that were not included in
this chart were insignificant because they had a weak
oscillator strength.
Below is a link to a UV-Vis Spectra of C
6H
5Cl.
Chlorobenzene
UV-Vis Spectra
The UV-Vis values that were calculated
are somewhat different from the accepted values. This is
most likely due to the errors due to approximations in the
calculations that were performed.
References:
1. Lide, D. R. CRC Handbook of
Chemistry and Physics; CRC Press: Boca Raton, 1992.
2. NIST:
National Institute of Standards and Technology. http://webbook.nist.gov/cgi/cbook.cgi?ID=C75467&Units=SI&Mask=80#IR-Spec
(accessed Mar 3, 2013).
Based on template by A. Herráez as modified by J. Gutow
Using directory /Users/student2/Desktop/Carly and Jesse/Web Site/C6H5Cl Website/C6H5Cl Jmol
adding JmolPopIn.js
...jmolApplet0
...adding Electrostatic_Potential.png
...adding Electrostatic_Potential.spt
...jmolApplet1
...adding Partial_Atomic_Charges.png
...adding Partial_Atomic_Charges.spt
...jmolApplet2
...adding Bond_Length_and_Angle.png
...adding Bond_Length_and_Angle.spt
...jmolApplet3
...adding 745_11_1_cm.png
...adding 745_11_1_cm.spt
...jmolApplet4
...adding 1119_39_1_cm.png
...adding 1119_39_1_cm.spt
...jmolApplet5
...adding 1600_66_1_cm.png
...adding 1600_66_1_cm.spt
...jmolApplet6
...adding 1473_04_1_cm.png
...adding 1473_04_1_cm.spt
...jmolApplet7
...adding 3403_50_1_cm.png
...adding 3403_50_1_cm.spt
...jmolApplet8
...adding Geometry_Optimization.png
...adding Geometry_Optimization.spt
...jmolApplet9
...adding HOMO.png
...adding HOMO.spt
...jmolApplet10
...adding LUMO.png
...adding LUMO.spt