Chlorine Monofluoride (FCl)
The MO Calculations for Chlorine Monofluoride (FCl)
For Chlorine Monofluoride, the
over all best calculated dipole moment was from AM1 calculation while
the best calculated dipole moment among the ab initio
theory calculation was 621G. The same pattern holds true for the bond
length. The 6-21G calculation yielded an 28.5% error for the dipole moment
and a 3.79% error for the bond length for Chlorine Monofluoride. The
data collected from the calculations is shown below.
Table 1: Dipole moment and bond length for the five levels of theory.
It is obvious that the results of the dipole moment and bond length from AM1 was the closest to the literature value. However, for ab initio theory, 6-21G had the closest results to the literature value. Therefore, we adopted 6-21G for the rest of the calculations for Chlorine Monofluoride. The geometry of chlorine fluoride under 6-21G, 6-31G, DZV, and AM1 are shown from Figure 1 to Figure 4. The bond length is labeled in each figure as well. The bond angle of FCl was not included since it is 180 degrees. | |||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||
|
The
Highest Occupied Molecular Orbital (HOMO) was found at orbital 13. The HOMO
was calculated by dividing the sum of the total number of electrons in
the molecule by 2. The Lowest Unoccupied Molecular Orbital (LUMO) was found at
orbital 14. The LUMO was the orbital that would be filled by the
excited valence electron(s) from the HOMO if proper excitation occurs.
| |||||||||||||||||||||||||||||||||||||||||
|
The
electrostatic potential of FCl using 6-21G calculation was shown in Figure 7.
The electrostatic potential used a color spectrum, which uses red to
indicate the lowest electrostatic potential and blue to indicate the
highest electrostatic potential. 7
The electrostatic potential shows the electron distribution in the molecules. 7
The partial atomic charge is shown in Figure 8. It is the distribution by the asymmetric scatter of the electrons in the molecule. Fluorine is more electronegative than chlorine. Therefore, the fluorine had the negative partial atomic charge; the electrons are more likely to hanging around fluorine. Since FCl is a diatomic molecule, there is only one kind of stretching possible. This is shown in Figure 9.
| |||||||||||||||||||||||||||||||||||||||||
|
| |||||||||||||||||||||||||||||||||||||||||
|
| |||||||||||||||||||||||||||||||||||||||||
|
The valence energy level diagrams corresponding to
the type of bonding occurring at specific levels is shown below in Table 2.
Table 2: Valence energy level diagrams corresponding to the type of bonding occurring at specific levels
| |||||||||||||||||||||||||||||||||||||||||
|
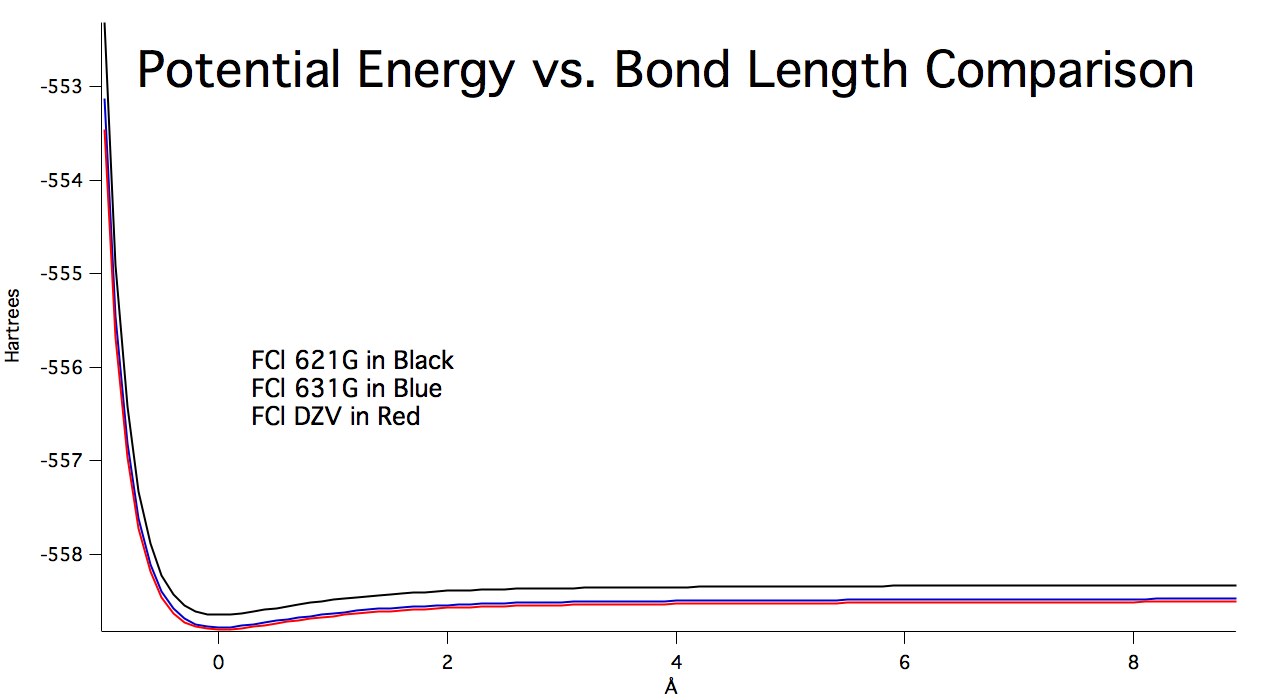
The following graph, Figure 10, shows the
relationship between potential energy and bond length using different
calculations. | |||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||
|
Figure 10: Potential energy curves at different levels of theory. The graph was generated by IGOR Pro. | |||||||||||||||||||||||||||||||||||||||||
|
| |||||||||||||||||||||||||||||||||||||||||
|
| |||||||||||||||||||||||||||||||||||||||||
|
|
Page skeleton and JavaScript generated by export to web function using Jmol 14.2.12_2015.01.22 2015-01-22 21:48 on Mar 9, 2015.