Picture so far:
(numbers next to atoms are their formal charges)
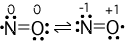
Example formal charge calculations
(first resonance structure): N-->5-3-(1/2)(4) = 0, O-->6 - 4 -
(1/2)(4) = 0. Since the first resonance structure has formal
charges closest to zero it is the preferred structure.