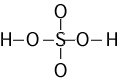
Note that H2SO4
requires a little more chemical knowledge than the others.
Knowing that S is the central atom is not enough. We need to know that
only the O's are bonded to the central S. The H's then need to be
attached to two of the O's. One way you might remember this is
that SO42- is one of the common polyatomic ions
that practicing chemists know about and most students are asked to
memorize. So you start by building the SO4 group and
then attaching the H's to the outside.
Picture so Far: