| Basis Set |
Bond Length |
| 3-21G |
1.432407 |
| 6-31G |
1.429702 |
| DZV |
1.416607 |
| Literature |
1.41451 |
| Number of diffuse orbital functions included in basis set |
3-21G | 6-31G | DZV |
| 0 | 1.322598 | 1.429702 | 1.500531 |
| 3 | failure | 1.146224 | 1.0979278 |
| 5 | failure | 0.978147 | 0.951230 |
| 7 | failure | 0.951641 | 0.970366 |
Table 2 - Dipole moments, Debye.
Literature Value: 0.827 Debye1The failures of the 3-21G basis set can likely be attributed to the insufficient size of the set. There is a general trend of more diffuse functions leading to more accurate results, with the exception of the DZV with 7 difuse functions. This is another example of how care must be taken when performing these calculations.
Below
is a graph of potential energy vs. bond stretch. In general it is
accepted that the larger the basis set, the lower the calculated energy
will be. However, these results are an exception to that trend,
as the DZV set is larger than the 6-31G set. Heavier atoms are
harder to deal with in computations, and this result may be explained
by an inability to accurately compute the properties of bromine.
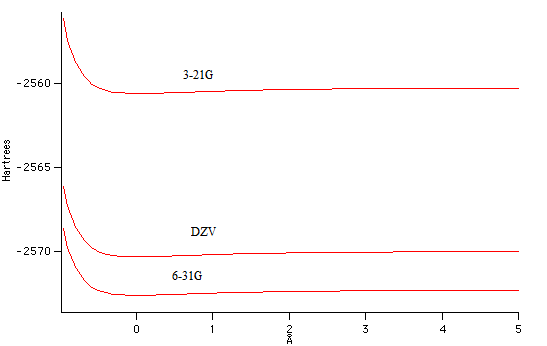
Figure
3 - Graph of potential energy vs. bond stretch. The larger basis
set DZV calculated a higher energy than the 6-31G basis set, which is
contrary to the expectation that larger basis sets produce lower
energies.
Below is a table of vibrational frequencies for HBr.
| 3-21G | 6-31G | DZV | Literature |
| 2491 | 2672 | 2654.5 | 26492 |
| 14.01 | 25052 | ||
| 13.55 | |||
| 4.17 |
with the largest basis set DZV coming the closest in this instance.
Below
is a graph of potential energy vs. bond stretch. In general it is
accepted that the larger the basis set, the lower the calculated energy
will be. However, these results are an exception to that trend,
as the DZV set is larger than the 6-31G set. Heavier atoms are
harder to deal with in computations, and this result may be explained
by an inability to accurately compute the properties of bromine.

Figure
3 - Graph of potential energy vs. bond stretch. The larger basis
set DZV calculated a higher energy than the 6-31G basis set, which is
contrary to the expectation that larger basis sets produce lower
energies.
(1) Lide, D. R., CRC Handbook of Chemistry and Physics, 77th Edition, CRC Press, 1996-1997.
(2) "UV-VIS Absorption Spectra of Gaseous Molecules and Radicals: Catalogue Spectra." Atmospheric Chemistry: Start Page. Web. 31 Mar. 2010. <http://www.atmosphere.mpg.de/enid/0,e0818c5350454b5452554d5f7375626b6174092d09093a095350454b5452554d
5f6b6174092d09487964726f67656e2b68616c69646573093a095350454b5452554d5f73746f66666e616d65092d09687
964726f67656e2b62726f6d696465093a095f7472636964092d093531363935/Spectra/Catalogue_5p4.html>.