|
|
| C2H4 Optimized Geometry |
According to these calculations, the most optimal geometry for C2H4 has a bond length of 1.33 Angstroms between the carbons and an angle of 116.4 degrees between the hydrogens on each carbon.
Lit comparison:For the literature3, an experimental value of 1.339 Angstroms between the carbons was found. As well as and experimental value of 117.6 degrees between the hydrogens on each carbon. The calculated values vary slightly from the experimental values, which is to be expected because the calculations are based on approximations, not exact values.
|
|
| C2H4 HOMO |
|
|
| Motion of C2H4 at 1110cm-1 |
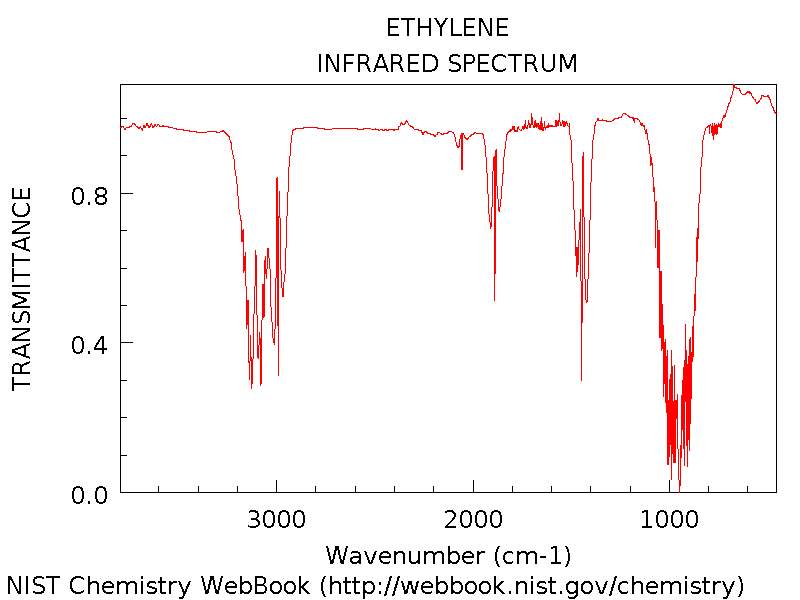
Vibrational frequencies are the
frequencies
at which a molecule vibrates. In the presence of Infrared
Radiation, the vibrations can be detected. The frequency of this
motion is 1110cm-1, and corresponds to the peak closest to
1000 above4.
|
|
| Motion of C2H4 at 1619cm-1 |
The frequency 1619cm-1
corresponds to the motion depicted on
the left, and corresponds to the peak shown second from the right above4.
|
|
| Motion of C2H4 at 1811cm-1 |
The frequency 1811cm-1
corresponds to the motion depicted on
the right, and corresponds to the peak shown near 1900 above4.
|
|
| Motion of C2H4 at 3335cm-1 |
The frequency 3335cm-1
corresponds to the motion depicted on
the left, and corresponds to the large peak furthest to the left above4.
|
|
| Motion of C2H4 at 3462cm-1 |
The frequency 3462cm-1
corresponds to the motion depicted on
the right, and corresponds to the large peak furthest to the left as
well4.