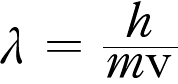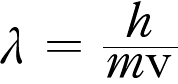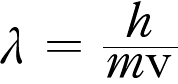Known Typos and Errors in Chem 105 Text
Spring 2004, first printing of Chemistry: The Science in Context
Compiled by Drs. Gutow, Matsuno and Mihalick with the help of Spring
2004, Fall 2004, Fall 2006 Chemistry 105 students.
Periodic
Table:
The atomic mass of Tellurium (#52) should be 127.60
Chapter 1:
The speed of light on pages 24 and 25 should be 2.99792458 x 108
m/s.
Page 21 equation 1.4
The denominator should be v (the
natural, unshifted frequency)
not v' (the shifted
frequency). The same typo appears in the equation shown in Sample
Exercise 1.3.
Page 28 table 1.3
1 mi/hr = 1.61 km/hr
Chapter 2:
Sample exercise 2.1 (page 60)
The masses of the neutron and proton do not match the values inside the
back cover.
For the practice exercise, using the tabulated values, the binding
energy is 9.0550 x 10-12 J.
Page 71 equation 2.12
first product is 144Cs (atomic number 55) not 144Ba
(atomic number 56)
Page 73 practice exercise 2.4
They have confused the mass numbers and atomic numbers. You can't
do the problem.
Page 74 equation 2.17
The atomic numbers should be 93 for Np and 94 for Pu.
Page 92 equation for t in left margin is missing a negative sign or
has the fraction within the ln inverted.
Should read either: t = {-t1/2/ln(2)}ln{At/Ao}
or {t1/2/ln(2)}ln{Ao/At}.
Page 98 problem 15: The given mass for 4He
includes electrons but the given mass for 1H does
not. Without the two electrons the 4He nucleus (an
alpha particle) weighs 4.00150 amu or the 1H mass including
electron is 1.007825 amu.
Page A-46 "Practice Tests" should read "Practice Exercises".
Page A-59 Answer to problem 9 should be 1.503 x 10-10 J
or 1.321 x 10-15 m
Page A-60 43d should read 0+1e since we have
lost one + charge. The other possibility is that this is a case
of
electron capture.
Chapter 3:
Page 155 in the review section the most useful form of the deBroglie
relation is the one in terms of the speed of a random particle not a
photon so the relation is better written as: 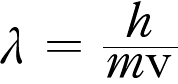
Page 156 the equation in problem 35 should be: 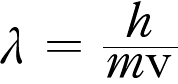
Page A-62 the answer to question 36 is not labelled (it's the
paragraph above 37).
Page A-63 the answer to question 79 is three elements:
the first configuration is for the ground state of Sc;
the second is the ground state of Ga;
the third is an excited state of Sc;
the fourth is an excited state of Cl.
Chapter 4:
Page 177: the last equation on the page should read
[(1 mol Na x 22.99 g/mol) + (1 mol Cl x 33.45 g/mol)] /mol NaCl=
not [(1 mol Na x 22.99 g/mol) X(1 mol Cl x 33.45 g/mol)] /mol NaCl=
Practice exercise 4.2 answer
PCl5 name should be phosphorus pentachloride and As2O3
should be diarsenic trioxide, based on the rules we know.
Phosphorus and arsenic can take on multiple charges so sometimes people
do name them using roman numerals to specify the charge on the positive
ion. As long as the charge on the negative ion is known, either
way clearly identifies the compound.
Problem 49. a. choices should be CO2 or N2O
Answers to problem 63: b and c are reversed.
Answer for problem 93.b: 6.55 g
Chapter 5:
Page 220: equation 5.3, which shows the relation between the
concentration of two dilutions, has an error.
Since the total moles of solute is constant, n= M*V in both
cases.
The relation should be MiVi=MfVf
, not MiVi=VfVf.
Answer to problem 53c should be 52.9 atm
Answers to problem 61: a. 299 g; b. 80.6 g; c. 177 g
Answer to problem 67 should be 0.0252 m
Answer to problem 73 should be Increase in boiling point goes as: 0.06
m FeCl3< 0.10 m MgCl2 < 0.20 m KCl since im
is biggest for KCl.
Chapter 6:
Page 305 & 314, Figures of NO3- resonance
structures. The right hand resonance structure should have a
double bond going to the top oxygen.
Page 314, example 6.9: the right hand figure needs a double bond to the
oxygen with a formal charge of zero.
Page 314, Answer to practice exercise: the first resonance structure
for NO2+ should have a triple bond on the right
hand side.
Chapter 7:
Page 347, in the figure at the bottom of the page the Lewis structure
for NO is wrong. It has too many bonds and electrons.
Table 7.2 on page 363 has the wrong number of bonding pairs for linear
and trigonal planar sets. The table on page 379 is correct.
Figure 7-27 has too many dashed lines. The erroneous and
corrected figures are shown below:
Wrong
|
Correct
|
|
|
Chapter 8:
Answer to Practice Exercise 8.11 is 0.0808 mole fraction O2
and 0.9920 mole fraction He.
Answer to Practice Exercise 8.14 is 9.5 x 10-5 mol/L.
The answer to Problem 27 is backwards. The question asks which
line is "not consistent"
thus the answer is curve #2.
End of Chapter Problem 87 the units of the requested answer should be
moles/L not moles/(L*atm).
Answer to End of Chapter Problem 129 should be 916 atm. Some
printings of the book have the answer wrong.
Chapter 9:
Page 444, Practice exercise 9.3, the energy change in the formation of
the O2- ion should be negative.
Chapter 10:
Page 475 The worked out calculation of NaCl density has some errors.
V = (5.628x10-8 cm)3 = 1.783x10-22 cm3
d = m/V = (3.882x10-22 g) / (1.783x10-22 cm3)
= 2.18 g/cm3
The answer to Problem 49 is 3.81 g/cm3
For problem 65, Cr2+ would have both choices but Cr3+
can have only one configuration.
Answers for Problem 71. V3+ has two unpaired
electrons, but high-spin versus low-spin is meaningless since there is
only one possible way of arranging the two d electrons as unpaired in
the lower energy d orbitals.
go to top of page
Last Update:5/9/06